Module 13: Water Quality and Characteristics of System Water in Aquaponic Systems
Summary / Description Text
This module examines the fundamental water quality parameters that govern the biological balance and overall performance of aquaponic systems. Since water serves as the shared medium for fish, plants, and nitrifying bacteria, even small variations in temperature, pH, dissolved oxygen, ammonia, nitrite, and nutrient concentration can directly influence system stability. The module explains how these parameters interact, how they affect the growth, metabolism, and health of each biological component, and why continuous monitoring is essential for sustainable system operation.
The module also discusses the nitrogen cycle as a core mechanism of aquaponics, describing the transformation of ammonia into nitrite and nitrate through nitrifying bacteria, the environmental conditions required for efficient conversion, and the impact of pH, temperature, alkalinity, and dissolved oxygen on this process. Additionally, the importance of hardness, alkalinity, water buffering capacity, and seasonal fluctuations is explored in the context of system reliability.
Furthermore, the module highlights the role of water quality management in supporting circular resource use and sustainable production. Proper water quality enhances nutrient recycling, reduces external fertilizer inputs, minimizes waste discharge, and increases operational efficiency. Water measurement tools (pH meters, DO meters, test kits) and analytical methods are introduced to provide learners with practical knowledge to assess and manage system conditions effectively.
Keywords
aquaponics, water quality, nitrogen cycle, dissolved oxygen, pH, alkalinity, hardness, nutrient availability, sustainable production, biological filtration
Learning Outcomes
After completing this module, learners will be able to:
• Understand the importance of water quality for fish, plants, and nitrifying bacteria in aquaponic systems.
• Identify key physical, chemical, and biological water quality parameters and explain their acceptable ranges.
• Describe the nitrogen transformation processes in aquaponics and factors affecting nitrification efficiency.
• Recognize the effects of pH, temperature, alkalinity, and dissolved oxygen on system stability and nutrient availability.
• Evaluate common water quality measurement tools and monitoring techniques used in aquaponic management.
• Explain how proper water quality management enhances sustainability, nutrient recycling, and overall production efficiency.
Presentation File
3.6. Water Quality And Characteristics Of System Water In Aquaponic Systems
Water quality in aquaponic systems is critically important for maintaining the healthy functioning of the biological cycle established among fish, plants, and bacteria. Since water serves as the shared living environment for all organisms in the system, even small changes in key parameters, such as temperature, pH, dissolved oxygen, ammonia, and nitrite, can directly affect overall system stability. When optimal water quality conditions are not maintained, fish may experience stress, reduced growth, and mortalities, while plants exhibit decreased nutrient uptake, stunted development, and lower yields (Somerville et al., 2014). Similarly, nitrifying bacteria lose their efficiency under low pH or oxygen levels, which can lead to the accumulation of ammonia at toxic concentrations (Antoniou et al., 1990). Therefore, continuous monitoring and proper management of water quality are essential to maintaining production efficiency as well as ensuring the sustainability and long-term stability of the system.
Moreover, maintaining water quality is highly important for the environmental and economic sustainability of aquaponic systems. When optimal conditions are ensured, fish waste is efficiently converted into plant nutrients, reducing the overall waste load and eliminating the need for additional fertilizers (Rakocy et al., 2006). This not only lowers production costs but also minimizes environmental impacts. Therefore, water quality is not merely a technical parameter that determines system performance; it is also a fundamental component that supports circular resource use and directly contributes to sustainable production goals.
Water quality in aquaponic systems is shaped by variations in physical, chemical, and biological parameters. Dissolved oxygen, pH, temperature, total nitrogen, and water hardness are the major water quality parameters within an aquaponic system (Somerville et al., 2014). The desired range of these parameters is determined according to the specific requirements of the fish, plants, and bacteria present in the system. At this point, defining the system’s water composition in accordance with the suitable ranges for each component is crucial for operational efficiency. Table 1 presents the recommended water quality parameter ranges for fish, plants, and nitrifying bacteria in aquaponic systems (Şekeroğlu et al., 2022; University of Latvia, 2023).
Table 1. Aquaponic system conditions (Şekeroğlu et al., 2022; University of Latvia, 2023)
|
Organism |
Temperature (°C) |
pH |
Ammonium (mg/L) |
Nitrite (mg/L) |
Nitrate (mg/L) |
Oxygen (mg/L) |
Example |
|
Warm-Water Fish |
22–32 |
6–8.5 |
<3 |
<1 |
<400 |
4–6 |
Tilapia, carp, perch |
|
Cold-Water Fish |
10–18 |
6–8.5 |
<1 |
<0.1 |
<400 |
6–8 |
Salmonids |
|
Plants |
16–30 |
5.5–7.5 |
<30 |
<1 |
— |
>3 |
Cabbages, basil, lettuce |
|
Bacteria |
14–34 |
6–8.5 |
<3 |
<1 |
— |
4–8 |
Nitrobacter, Nitrosomonas, etc. |
When Table 1 is examined, it is evident that temperature and dissolved oxygen are the parameters that vary most significantly among fish species depending on their specific requirements. For example, in warm-water species such as tilapia, carp, and sea bass, an optimal temperature of 22–32 °C and dissolved oxygen levels of 4–6 mg/L are desired. In contrast, cold-water species like salmon require lower temperatures of 10–18 °C and higher dissolved oxygen levels of 6–8 mg/L. For plants in aquaponic systems, the recommended water quality conditions include a minimum dissolved oxygen concentration of ≥3 mg/L, a pH range of 5.5–7.5, a temperature range of 16–30 °C, ammonium levels below 30 mg/L, and nitrite levels below 1 mg/L.
The water temperature required for plants varies depending on the crop species selected and the climatic conditions of the location where the system is established. For instance, a temperature range of 23–28 °C is suitable for temperate-climate plants such as cabbage and basil, whereas cool-climate crops like lettuce grow better at water temperatures between 17–25 °C (University of Latvia, 2023). For bacteria, the recommended water quality conditions include a temperature range of 14–34 °C, a pH range of 6–8.5, dissolved oxygen levels of 4–8 mg/L, ammonium concentrations below 3 mg/L, and nitrite concentrations below 1 mg/L. In particular, for efficient nitrification by Nitrobacter and Nitrosomonas, key bacteria in the nitrogen cycle, the system must maintain a temperature range of 25–30 °C and a pH range of 7–9 (Antoniou et al., 1990). Additionally, optimal temperature ranges are reported as 25–29 °C for Nitrobacter and 21–29 °C for Nitrosomonas (University of Latvia, 2023). Figure 1 presents a schematic illustration of how aquaponic system water parameters affect each system component (Wongkiew et al., 2017).
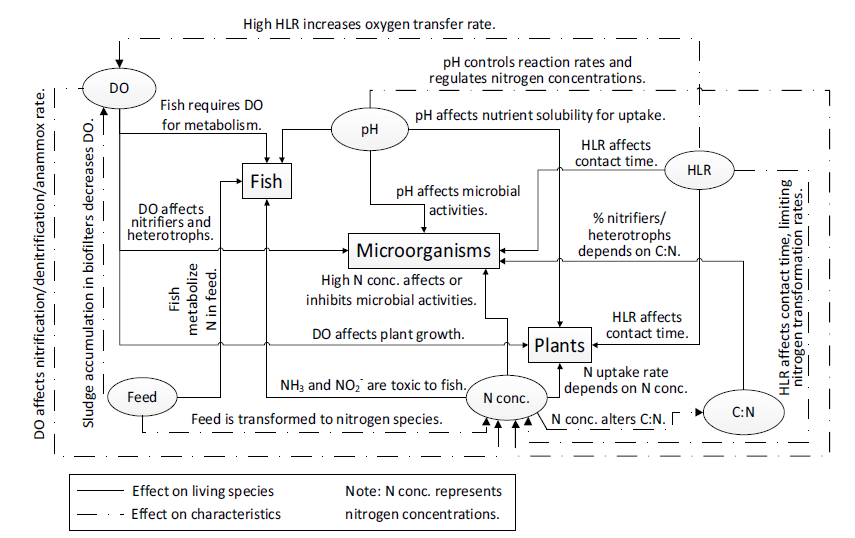
Figure 1. Effects of aquaponic system water parameters on system components (Wongkiew et al., 2017)
When the flow of aquaponic system water parameters in Figure 1 is examined, it is evident that increases or decreases in these parameters have different effects on fish, plants, and bacteria. For example, dissolved oxygen directly influences bacterial activity, fish metabolism, and plant growth; pH controls reaction rates within the system, regulates nitrogen concentrations, affects nutrient solubility for biological uptake, and therefore influences microbial activity. Additionally, the carbon-to-nitrogen (C:N) ratio determines the relative presence of nitrifying bacteria versus heterotrophic bacteria operating within the system.
3.6.1. Importance of Dissolved Oxygen Parameter in Aquaponic System Water
In aquaponic systems, dissolved oxygen is expressed as the amount of molecular oxygen (in mg) present per liter of water (Somerville et al., 2014). All living organisms require dissolved oxygen for survival. In aquaponic systems, a dissolved oxygen concentration above 5 mg/L is considered optimal for both the fish tank and the inlet of the plant growing bed (Bernstein, 2011; Rakocy, 2007; Somerville et al., 2014). If dissolved oxygen levels fall below the optimal threshold in the fish tank, fish mortality can occur. However, some fish species are capable of surviving even at low dissolved oxygen concentrations. For instance, hardy species such as tilapia have been reported to tolerate conditions where dissolved oxygen levels drop below 1 mg/L (El-Sayed, 2006).
Dissolved oxygen is also required for the nitrification process to occur. In this context, it is reported that a dissolved oxygen concentration above 1.7 mg/L must be maintained in biofilters (Ruiz et al., 2003). Dissolved oxygen directly influences plant growth as well (Wongkiew et al., 2017). The dissolved oxygen required in the system is supplied by air pumps. Figure 2 presents a chart illustrating the effect of dissolved oxygen levels in system water on fish (University of Latvia, 2023).
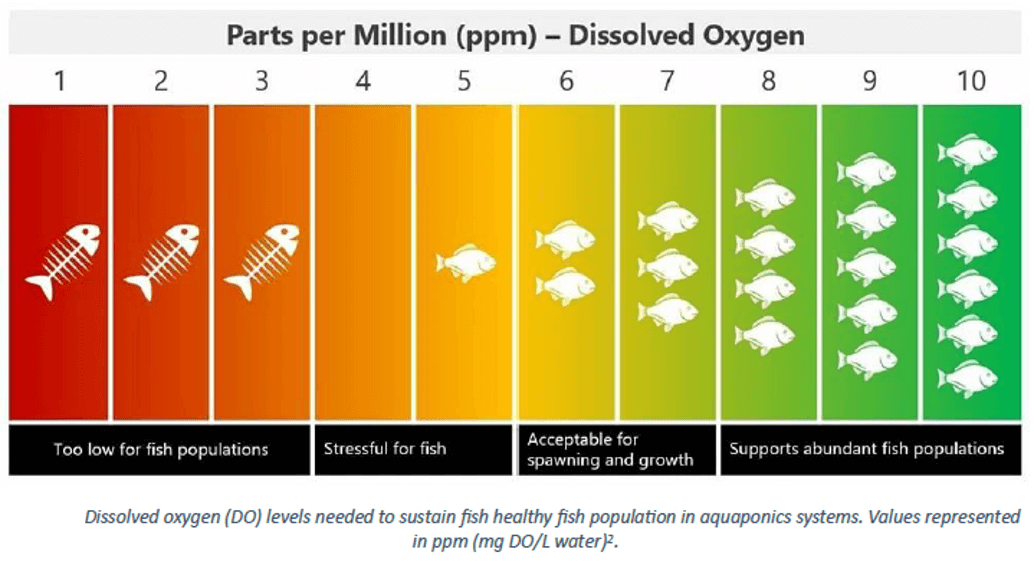
Figure 2. Effects of dissolved oxygen levels in aquaponic system water on fish (University of Latvia, 2023)
3.6.2. Importance of pH Parameter in Aquaponic System Water
In aquaponic systems, pH is important for fish metabolism, microbial activities, and the ability of plants to assimilate nitrogen (Wongkiew et al., 2017). The pH of the system water plays a key role in regulating the solubility of minerals such as calcium, magnesium, and phosphorus, thereby maintaining the nutrient uptake dynamics of plants (Resh, 2013). At this point, pH management is a parameter that directly affects the availability of nutrients for plants. While a pH range between 6.5 and 7.5 is a suitable range for fish, it is reported that some plants have difficulty absorbing certain nutrients in this pH range (Magalhães et al., 2025). The pH level of system water is also critical for the nitrification process. Since nitrifying bacteria function inefficiently at pH levels below 6, low pH conditions reduce biofiltration capacity, leading to elevated ammonia concentrations and creating stressful and unstable conditions for the other organisms in the system (Somerville et al., 2014).
In addition, the CO₂ released through fish respiration reacts with the system water to form carbonic acid (H₂CO₃), which lowers the pH and leads to the formation of an acidic environment (Wongkiew et al., 2017). Since a pH level close to neutral is preferred for all living organisms in the system, basic compounds such as potassium hydroxide (KOH), calcium hydroxide (Ca(OH)₂), and calcium bicarbonate (Ca(HCO₃)₂) can be used to adjust the system pH to the desired levels (Rakocy et al., 2003; Wongkiew et al., 2017). Figure 3 summarizes the effect of pH levels in system water on the availability of nutrients such as nitrogen, phosphorus, potassium, and magnesium (University of Latvia, 2023).
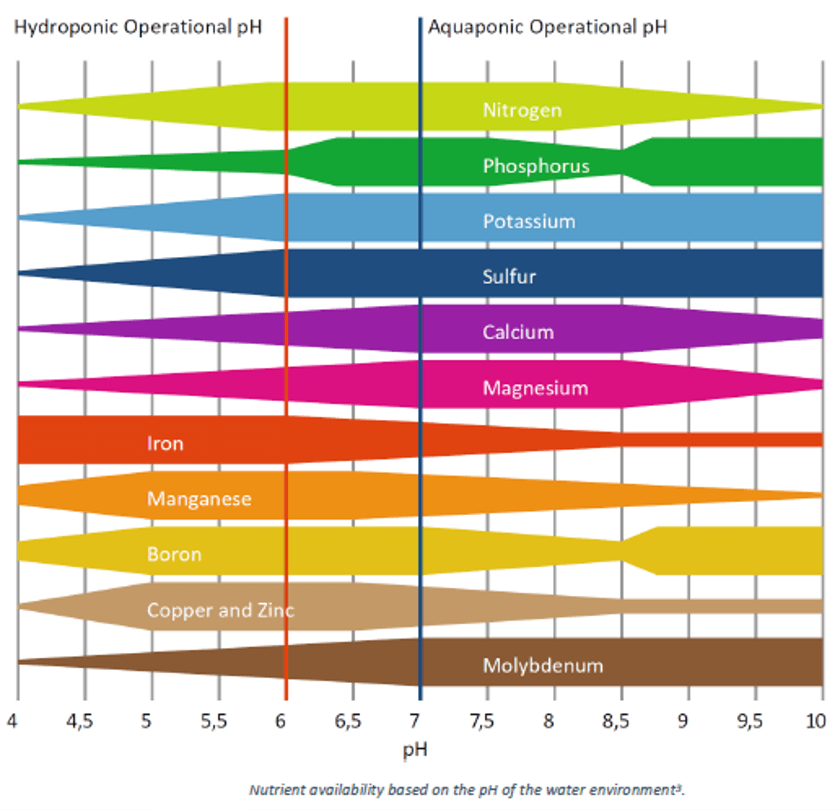
Figure 3. Effect of system water pH levels on nutrient availability (University of Latvia, 2023)
In aquaponic systems, the lack of nutrients plants need to absorb is a critical problem affecting the entire system. It has been reported that the recycling of fish waste into the plant system provides most of the nutrients plants need, but elements such as iron, potassium, and calcium often require supplementation (Magalhães et al., 2025).
3.6.3. Importance of Temperature Parameter in Aquaponic System Water
Water temperature is a key parameter that influences the amount of dissolved oxygen in the system, calcium uptake by plants, and the toxicity level of ammonia (University of Latvia, 2023; Somerville et al., 2014). The water temperature of the system is highly important for achieving rapid growth rates, efficient feed conversion, and minimizing disease occurrence (University of Latvia, 2023). Therefore, the water temperature in an aquaponic system must be maintained at a level suitable for both the fish and the plants selected for production. It is reported that seasonal changes in the system can affect fish metabolism, bacterial activity and nutritional needs of plants (Magalhães et al., 2025).
However, adjusting water temperature requires energy input, which can increase operational costs in aquaponic systems (Somerville et al., 2014). It is generally recommended that aquaponic system water temperatures remain between 18–30 °C, as this range is suitable for all system components (University of Latvia, 2023). To minimize temperature fluctuations that may occur throughout the day, shading structures should be used to protect the water surface from direct sunlight (Somerville et al., 2014).
3.6.4. Importance of Total Nitrogen Parameter in Aquaponic System Water
Nitrogen is an essential element for living organisms and enters the aquaponic system primarily through the proteins in fish feed. The portion of nitrogen that is not utilized by the fish is excreted into the water mainly in the form of ammonia (Somerville et al., 2014). In aquaponic systems, the nitrogen contained in fish waste is converted by bacteria into forms that are usable by plants (Kim et al., 2005). This conversion is indispensable for the functioning of aquaponic systems. The nitrogen forms that plants can assimilate are nitrite (NO₂⁻) and nitrate (NO₃⁻). Ammonia and nitrite are approximately 100 times more toxic than nitrate and pose a significant threat to fish health (Somerville et al., 2014). Sudden temperature shifts, pH fluctuations, and low alkalinity can negatively affect nitrifying bacteria, leading to imbalances in the nitrogen cycle (Magalhães et al., 2025). It is reported that four key water quality parameters influence nitrogen transformation in aquaponic systems (Wongkiew et al., 2017). These are pH, dissolved oxygen, hydraulic loading rate, and the carbon-to-nitrogen (C:N) ratio. In a healthy aquaponic system, ammonia and nitrite levels are typically maintained between 0–1 mg/L (Somerville et al., 2014). Nitrogen transformation in aquaponic systems is summarized in Figure 4 (Wongkiew et al., 2017).
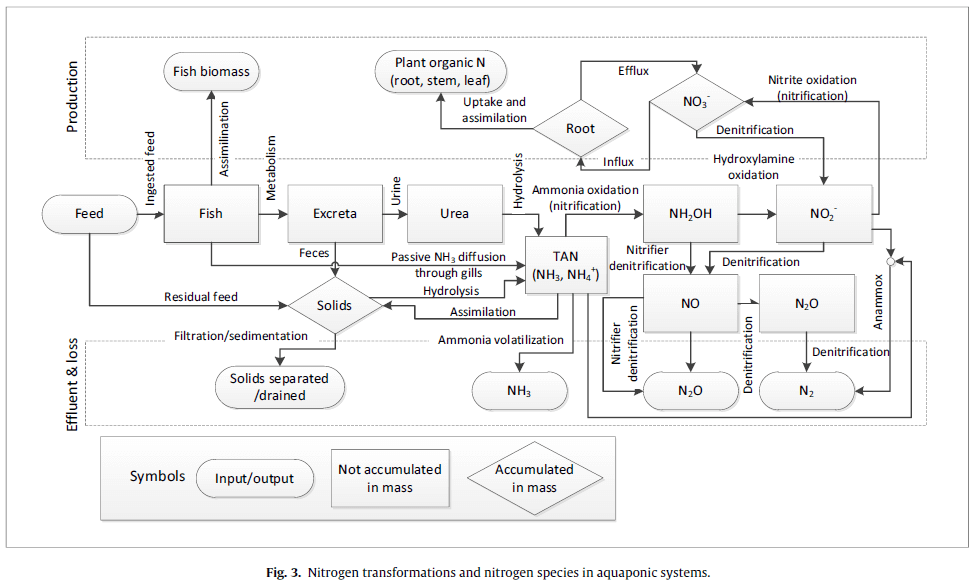
Figure 4. Nitrogen transformation in aquaponic systems (Wongkiew et al., 2017)
3.6.5. Importance of Hardness Parameter in Aquaponic System Water
Hardness is classified into two types: carbonate hardness and total hardness. Total hardness refers to the concentration of calcium (Ca²⁺) and magnesium (Mg²⁺) ions in the water, while carbonate hardness represents the concentration of dissolved carbonate (CO₃²⁻) and bicarbonate (HCO₃⁻) compounds, expressed as mg/L CaCO₃ (Somerville et al., 2014). For aquaponic systems, carbonate hardness is highly important because dissolved carbonate and bicarbonate ions buffer pH fluctuations and prevent sudden acidification of the system (University of Latvia, 2023). If the hydrogen ions released during nitrification are not buffered, the pH can rapidly drop, disrupting the system’s stability and functioning; therefore, maintaining adequate levels of carbonate and bicarbonate is essential. At this point, alkalinity plays a critical role. The buffering capacity, or alkalinity, reflects the water’s ability to maintain a stable pH (Klinger-Bowen et al., 2011).
In addition, the calcium and magnesium contained within total hardness function as micronutrients for plants and are important for metabolic processes in fish (University of Latvia, 2023; Chandramenon et al., 2024). Table 2 presents the water hardness levels according to the calcium carbonate content of system water (Somerville et al., 2014). In aquaponic systems, the optimal total hardness level, representing soft to moderately hard water (the sum of total and carbonate hardness), is reported to be between 60–140 mg/L (Chandramenon et al., 2024).
Table 2. Water hardness levels based on the concentration of calcium carbonate (Somerville et al., 2014).
|
Water hardness level |
Concentration (mg/L) |
|
Soft |
0-60 |
|
Moderately hard |
60-120 |
|
Hard |
120-180 |
|
Very hard |
>180 |
Deterioration of water quality in aquaponic systems creates a cascade of negative effects on fish, plants, and nitrifying bacteria. Increases in water temperature, decreases in dissolved oxygen levels, accumulation of ammonia and nitrite, and low pH conditions can lead to severe stress, loss of appetite, gill damage, and mortality in fish; while in plants, these conditions may cause nutrient deficiencies, leaf chlorosis, root rot, and stunted growth (Somerville et al., 2014; Chandramenon et al., 2024). Declining water quality also reduces the efficiency of nitrifying bacteria, resulting in uncontrolled increases in ammonia and nitrite levels and disrupting the overall stability of the system. In the long term, poor water quality can lead to serious problems such as increased disease risks and reduced production efficiency (Somerville et al., 2014).
3.6.6.Measurement and Monitoring of Aquaponic System Water Quality Parameters
Water parameters in aquaponic systems are regularly monitored to ensure that they remain within the desired ranges. In this context, water quality is routinely assessed using measurement devices such as pH meters, DO meters, and test kits, allowing for early intervention in case of potential problems (Klinger-Bowen et al., 2011). The proper and combined use of these instruments ensures that aquaponic systems operate in a sustainable and stable manner. Figure 5 presents images of various water quality measurement devices used in aquaponic systems (Klinger-Bowen et al., 2011).

Figure 5. Images of Water Quality Measurement Devices Used in Aquaponic Systems (Klinger-Bowen et al., 2011)
Water quality parameters in aquaponic systems are monitored using different methods. For example, hardness is measured using the EDTA titrimetric method, while ammonia is analyzed using the Colorimetric Nesslerization Method (Ebeling & Timmons, 2010). Table 3 summarizes the measurement methods for various water quality parameters (Ebeling & Timmons, 2010).
Table 3. Monitoring methods for selected water quality parameters in aquaponic system water (Ebeling & Timmons, 2010).
|
Water Quality Parameters |
Monitoring Methods |
|
pH |
pH Meters |
|
Ammonia |
NH3 Nitrogen (Ammonia) Colorimetric Nesslerization Method: nessler-ammonia reaction produces a yellow to brown color, measured with spectrophotometer at 425 nm. |
|
Nitrite |
4500-NO2″ Nitrogen (Nitrite) Colorimetric method with diazotized sulfanilamide with NED dihydrochloride creating a reddish purple azo dye measure with a spectrophotometer at 543 nm. |
|
Nitrate |
45OO-NO3″ Nitrogen (Nitrate) Cadmium reduction to nitrite and measurement of NO2″. |
|
Hardness |
EDTA titrimetric method using EDTA and ethylenediaminetetraacetic acid and Calmagite. Titrated with EDTA from a red to a blue color. |
|
Temperature |
Temperatures are traditional made with mercury-filled thermometers, but non-mercury or electronic temperature meters are preferred due to the toxicity of mercury in the environment. |
Maintaining water quality in aquaponic systems is critical for fish health, plant nutrient uptake, and the efficient functioning of nitrifying bacteria. Therefore, the system continuously supports the physical, chemical, and biological balance of the water through various equipment and materials. Mechanical filters and settling tanks remove solid waste from the water, while biofilters provide the necessary surface area for bacteria that convert ammonia to nitrite and nitrite to nitrate (Bregnballe, 2015). Air pumps, diffusers, and air stones increase dissolved oxygen levels, helping maintain the oxygen balance required by both fish and bacteria (Somerville et al., 2014).
References
Antoniou, P., Hamilton, J., Koopman, B., Jain, R., Holloway, B., Lyberatos, G., & Svoronos, S. A. (1990). Effect of temperature and pH on the effective maximum specific growth rate of nitrifying bacteria. Water Research, 24(1), 97-101.
Bernstein, S., 2011. Aquaponic Gardening: A Step-by-Step Guide to RaisingVegetable and Fish Together. New Society Publishers, Grabiola, Canada.
Bregnballe, J. (2015). A guide to recirculation aquaculture: An introduction to the new environmentally friendly and highly productive closed fish farming systems (3rd ed.). Food and Agriculture Organization of the United Nations (FAO) & Eurofish International Organisation.
Chandramenon, P., Aggoun, A., & Tchuenbou-Magaia, F. (2024). Smart approaches to Aquaponics 4.0 with focus on water quality – Comprehensive review. Computers and Electronics in Agriculture, 225, Article 109256.
Ebeling, J. M., & Timmons, M. B. (2010). Recirculating aquaculture. Ithaca, NY, USA: Cayuga Aqua Ventures.
El-Sayed, A.F.M., 2006. Tilapia Culture. CAB eBooks, Oceanography Department, Faculty of Science, Alexandria University, Alexandria, Egypt. https://doi.org/10.1079/9780851990149.0000.
Kim, D.-J., Ahn, D.H., Lee, D.-I., 2005. Effects of free ammonia and dissolved oxygen on nitrification and nitrite accumulation in a biofilm airlift reactor. Korean J. Chem. Eng. 22, 85–90.
Klinger-Bowen, R. C., Tamaru, C. S., Fox, B. K., McGovern-Hopkins, K., & Howerton, R. (2011). Testing your aquaponic system water: A comparison of commercial water chemistry methods. College of Tropical Agriculture and Human Resources, University of Hawai‘i at Mānoa.
Magalhães, T. B., Silva, N. C. S., Souza, R. J. F., Luz, R. K., von Brand, E., Merino, C., & Merino, G. E. (2025). Cultivo integrado de truta arco-íris com alface rumo a uma agroaquicultura sustentável. Aquaculture Brasil, Edição Anual, 1–8.
University of Latvia. (2023). Water quality in aquaponics (D.3.2.1 – Training materials on water quality). In TRANSborder cooperation for circular soil-less FARMing systems (TransFarm) project report. Interreg Central Baltic Programme (Project CB0100007)
Rakocy, J. E., Masser, M. P., & Losordo, T. M. (2006). Recirculating aquaculture tank production systems: Aquaponics—Integrating fish and plant culture. Southern Regional Aquaculture Center (SRAC) Publication No. 454.
Rakocy, J., Shultz, R.C., Bailey, D.S., Thoman, E.S., 2003. Aquaponic production oftilapia and basil: comparing a batch and staggered cropping system. SouthPacific Soil. Cult. Conf. 648 South Pacific Soil, 63–69.
Rakocy, J., 2007. Ten guidelines for aquaponic systems. Aquaponics J. 3rd Quarte,14–17.
Resh, H.M., 2013. Hydroponic Food Production: A Definitive Guidebook for theAdvanced Home Gardener and the Commercial Hydroponic Grower, 7th ed.CRC Press, Northwestern.
Ruiz, G., Jeison, D., Chamy, R., 2003. Nitrification with high nitrite accumulation forthe treatment of wastewater with high ammonia concentration. Water Res. 37,1371–1377.
Somerville, C., Cohen, M., Pantanella, E., Stankus, A. & Lovatelli, A. (2014). Small-scale aquaponic food production.Integrated fish and plant farming. Rome: FAO Fisheries and Aquaculture Technical Paper. No. 589.
Şekeroğlu, A., Kökten, K., & İnci, H. 2022. Tarım ve hayvancılığın sürdürülebilirlik dinamikleri üzerine akademik çalışmalar. Ankara: İKSAD Publishing House.
Wongkiew, S., Hu, Z., Chandran, K., Lee, J.W., Khanal, S.K., 2017. Nitrogen transformations in aquaponic systems: a review. J. Aquac. Eng. Fish. Res. 76, 9–19.